Comparative assessment of the ability of enterosorbents to bind protein substances
I. I. Gerashchenko, T. V. Krupskaya, V. V. Turov Institute of Surface Chemistry named after A.A. Chuyko, NAS of Ukraine,
Kyiv, st. Generala Naumov, 17
Comparative evaluation of the ability of enterosorbents to bind protein substances
Resume. A comparative assessment of the sorption activity of representatives of various classes of enterosorbents in relation to high-molecular protein compounds was carried out. As a marker of the effectiveness of the sorbents, the indicator of the sorption activity of such protein substances as human serum albumin (SAH) and gelatin was used. Samples of enterosorbents, the most widely represented on the market of Ukraine - Atoxyl, Smekta, Enterosgel, activated carbon - were studied by the methods of thermogravimetry, microscopy and adsorption of proteins in the aqueous medium. The maximum sorption activity of Atoxyl has been proven at a pH corresponding to the medium of the upper part of the small intestine, including in terms of a therapeutic dose.
Resume. A comparative assessment of the sorption activity of representatives of different classes of enterosorbents with respect to high-molecular protein compounds was carried out. As a marker of the effectiveness of the sorbents, the sorption activity index of such protein substances as human serum albumin (SAL) and gelatin was used. By the methods of thermogravimetry, microscopy and adsorption of proteins in an aqueous medium, samples of enterosorbents most widely represented on the Ukrainian market - Atoxil, Smecta, Enterosgel, activated carbon were studied. The maximum sorption activity of Atoxil was proven at a pH corresponding to the contents of the upper small intestine, including in terms of the therapeutic dose.
Summary. There was carried out a comparative evaluation of the sorption activity of different classes of enterosorbents relative to high molecular weight protein substances. As a marker of the sorbents efficiency action, the sorption activity index of proteins such as human serum albumin (HSA) and gelatin was used. The methods of thermogravimetry, microscopy and protein adsorption in water medium were used to study the samples of the most widely represented enterosorbents in the Ukrainian market, such as: Atoxil, Smecta, Enterosgel, and Activated Charcoal. The maximum sorption activity of Atoxil in terms of the therapeutic dose under the pH, which corresponds to the medium of the upper section of the small intestine, was proven.
At present, enterosorption, as a method of removing foreign substances from the body, falling from the environment or formed in the body itself as a result of pathological processes, is widely used in medical practice. Enterosorbents have shown their effectiveness in the treatment of diarrhea syndrome, food poisoning, bacterial infections, allergic reactions and other pathological processes accompanied by intoxication [1, 2].
The pharmaceutical market of Ukraine presents sorbents of various classes: based on highly dispersed silicon dioxide (Atoxyl), methylsilicic acid hydrogel (Enterosgel), montmorillonite clay (Smecta) and various forms of activated carbon [3, 4]. Representatives of different classes of enterosorbents can differ significantly in their sorption activity and ability to bind toxin molecules of different molecular weight. From a practical point of view, a comparative study of the effectiveness of the use of different classes of enterosorbents is of significant interest.
As is known, the basis of the pathogenesis of bacterial infections and other diseases accompanied by intoxication is the accumulation in the body of pathological proteins, bacterial toxins or oligomeric protein complexes, which are substances with a high molecular weight. The molecular mass (m.m.) of typical protein toxins is 15–170 kDa. So, for example, M.m. botulinum toxin is 150 kDa [5]. Since a significant part of toxins accumulating and forming in the gastrointestinal tract is associated with protein molecules or has a protein origin, it seems necessary to evaluate the sorption activity of representatives of various classes of enterosorbents in relation to high molecular weight protein compounds. As a marker of the activity of sorbents, the efficiency of the sorption of such protein substances as human serum albumin (SAA) and gelatin [1] can serve. Gelatin is a protein that is a mixture of polymer homologues with a molecular weight that corresponds to the typical weight of most protein toxins. In particular, the adsorption of gelatin is introduced as a parameter for assessing the quality of adsorbents in pharmacopoeial methods.
Materials and methods
Samples of enterosorbents of the corresponding series were taken for research (Table 1).
Table 1.

As test substances for the study of protein adsorption, we used: 1) 10 % solution of human serum albumin (solution for infusions, medicinal product); 2) edible gelatin Bloom 200 (Belgium), series 1574737. The test was carried out by the method of constructing an adsorption isotherm according to the methodological recommendations of the State Expert Center of the Ministry of Health of Ukraine [6]. Weight doses of drugs were the same in terms of dry residue, the content of which was determined using thermal analysis.
Results and discussion
Thermogravimetric studies were carried out on a Paulik&Paulik derivatograph. The sample heating rate was 10 degrees/min, the limit temperature in the furnace was 1000 °C. The measurements were carried out to determine the amount of water and auxiliary substances in the samples. At the same time, we used the fact that water is removed from systems with a developed surface, to which the studied samples belong, at temperatures T < 150 °C. At temperatures T < 400 °C, the gasification of most organic compounds (sugar, polymers) used in some enterosorbents as auxiliary substances occurs. Alkyl groups attached to the surface of organosilicon compounds can be destroyed at temperatures up to 550 °C. Thermograms of the studied samples are shown in Fig. 1a-g. In particular, mass loss curves (TG curve), as well as DTG and DTA curves are presented. The DTG curve is a dm/dT derivative. It makes it possible to more accurately determine the position of the inflection points on the mass loss curve, which corresponds to the processes that occur in the sample when the temperature increases. The DTA curve is the second derivative of mass loss. With its help, you can identify the type of process. Thus, maxima on the DTA curve correspond to exothermic processes, and minima to endothermic processes.
For the preparation of dioctahedral smectite (Fig. 1a) at T < 150 °C, the mass of the sample practically does not change, which indicates the almost complete absence of bound water. Two minima are recorded on the DTG curve at 180 and 260 °C. The inflection points on the mass loss curve (TG curve) correspond to them. The maximum mass loss of the sample is 27 % at 700 °C. According to the manufacturer's instructions, the main substance is dioctahedral smectite (3 g), and the auxiliary substances are glucose - 0.749 g, sodium saccharin - 0.007 g, and vanillin - 0.004 g. Thus, the loss of mass during heating of the sample occurs due to the destruction of auxiliary substances, and their amount corresponds to the one declared by the manufacturer. The presence of one broad maximum on the DTA curve indicates the exothermicity of the processes of destruction of auxiliary substances (heat is released when heated).
Unlike dioctahedral smectite, during thermal destruction of activated carbon, gasification of all carbon material present in the sample occurs. The ash content of the sample is 9.05 % (Fig. 1b). The first minimum on the DTG curve is observed at 100 °C. It corresponds to the evaporation of the water present in the sample, the amount of which is 7 % by weight. The second minimum at 280 °C corresponds to the destruction of polymer substances, the amount of which, according to the TG curve, is about 12 % mass. Since auxiliary substances are not specified by the manufacturer, the most likely is the presence of a polymer component used for the production of tablet forms. At 330 °C, an endothermic peak is observed on the DTA curve. It can be caused by the rearrangement of the carbon skeleton of polymer molecules during their carbonization.
The ash content of the methylsilicic acid hydrogel sample (Fig. 1c) is 6.7 % mass. This is the amount of dry residue that was formed when polymethylsiloxane was heated. About 93 % of the mass of the sample is water, which is used by the manufacturer for the production of hydrogel and is removed from the preparation at T < 150 °C. Accordingly, one minimum is registered on the DTG curve at 100 °C. A small endothermic maximum on the DTA curve at 500 °C can be associated with the removal of hydroxyl groups from the polymethylsiloxane matrix, which occurs together with the destruction of methyl groups. Thus, the amount of the active substance in the methylsilicic acid hydrogel sample does not exceed 6–7 % mass.
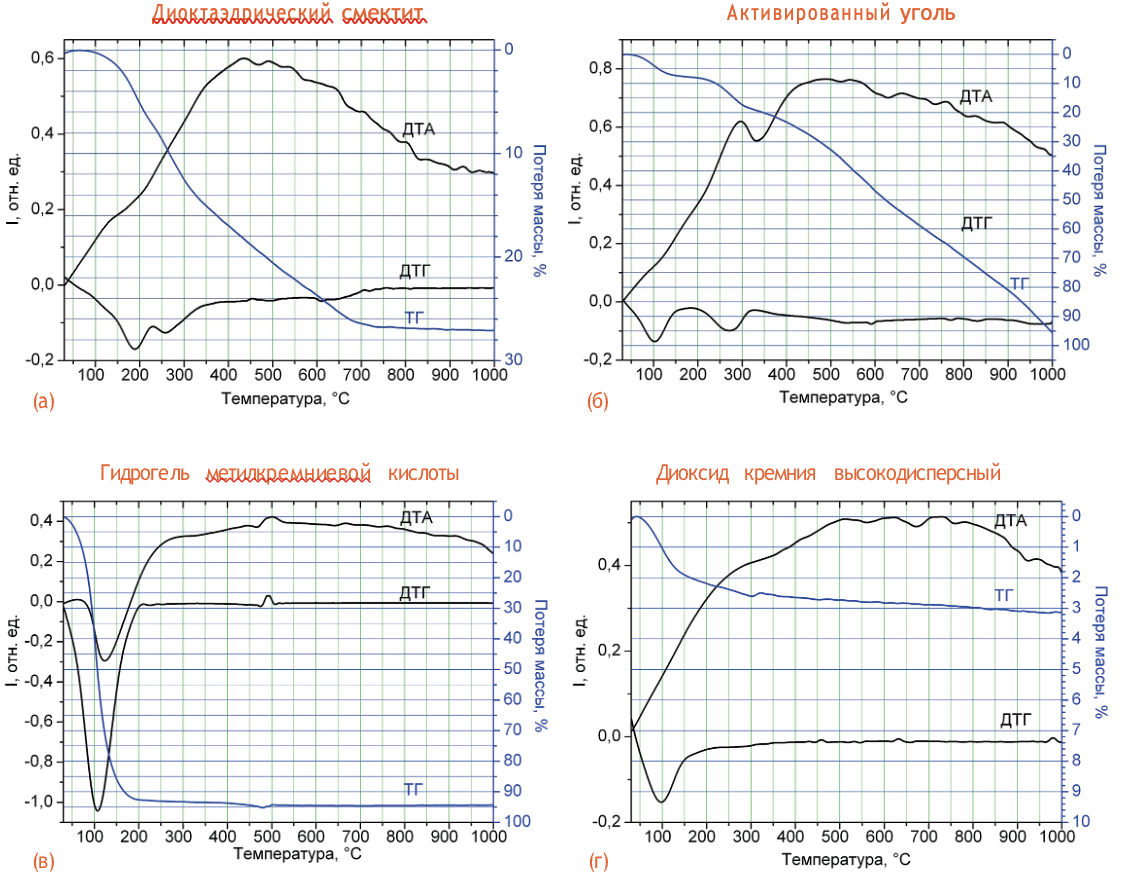
Rice. 1. Thermograms of the investigated sorbents: dioctahedral smectite – (a); activated carbon – (b); methylsilicic acid hydrogel – (c); highly dispersed silicon dioxide - (g).
The thermogram of a sample of highly dispersed silicon dioxide is shown in Fig. 1 year It shows one minimum on the DTG curve at 100 °C, which corresponds to mass loss due to moisture evaporation. The initial moisture content of the sample is 3 % mass, which corresponds to the standards laid down in the Pharmacopoeia and is consistent with the quality control standards of the manufacturer. Small exothermic peaks at 500–800 °С probably correspond to the process of condensation of silanol groups.
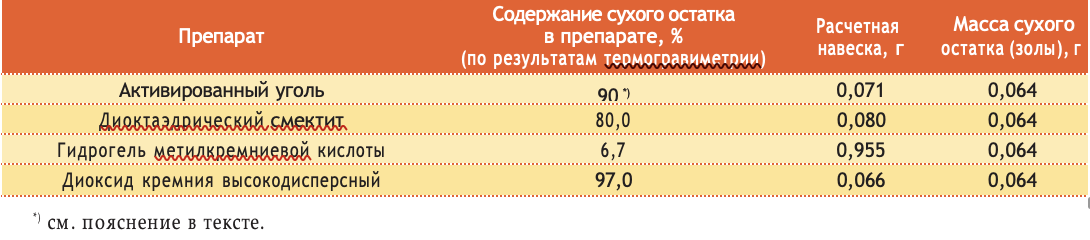
Summary indicators of the content of dry residue after calcination of samples of the investigated sorbents at 1000 °C are presented in table. 2.
Table 2.
The content of the dry residue after calcination of the samples of the investigated sorbents at 1000 °C

Photomicrographs of sorbents obtained with a Primo Star, Zeiss microscope are presented in fig. 2.

Rice. 2. Photomicrographs of sorbents obtained with the Primi Star Ceis microscope, Yena: dioctahedral smectite – (a); activated carbon - (b); methylsilicic acid hydrogel – (c); highly dispersed silicon dioxide - (g).
The studied materials, with the exception of methylsilicic acid hydrogel, are present in the form of particles, the size of which ranges from units to several tens of microns. A sample of methylsilicic acid hydrogel was studied in the form of an aqueous gel, since its drying leads to a complete change in morphology and surface characteristics. Optically inhomogeneous structures are observed on the surface of the gel (Fig. 2c), which is probably due to the crystallization of the gel, the spontaneous change of the dispersion medium, and the separation of the liquid phase.
When studying the activity of protein sorption, 8 ml of solutions of SAC or gelatin with increasing concentration were added to 7 samples of each of the preparations. For the comparability of the results, the obtained adsorption data for dioctahedral smectite, hydrogel of methylsilicic acid, and highly dispersed silicon dioxide were calculated for the same mass of dry residue, determined by the method of thermal analysis (see above). Since activated carbon is completely gasified during thermal analysis, the weight of the sample was calculated based on the composition of the tablet declared by the manufacturer (Table 3).
Calculation of a portion of the drug taken to study protein sorption activity
Table 3.
Water solutions of SAC and gelatin were used for the study. The concentration of sorbent solutions (Co) added to samples was in the range of 0–1 % (0–10 mg/ml) for SAC and in the range of 0–0.8 % (0–8 mg/ml) for gelatin. Test tubes with the obtained suspensions were shaken on a shaking apparatus for 2 hours, then centrifuged at a frequency of 6000 rpm for 20 minutes. 4 ml of biuret reagent was added to 1 ml of each centrifuge, mixed and left for 30 min.
The optical densities of the obtained solutions and the reference solution were measured at a wavelength of 540 nm against a blank solution. To prepare the reference solution, 4 ml of biuret reagent was added to 1 ml of the standard protein solution, mixed and left for 30 minutes. The blank solution was a mixture of 1 ml of water and 4 ml of biuret reagent. Based on the linear nature of the dependence of the optical density on the concentration, the concentration of the protein in the centrifuge (equilibrium concentration, Cr) was found. Then the amount of adsorption (A, mg/g) was calculated according to the formula:
A = (Со – Ср) × 125,
where 125 (coefficient) = 8 ml of medium/0.064 g weight dose.
The adsorption isotherms of gelatin and SAC on the surface of highly dispersed silicon dioxide are shown in Fig. 3. Taking into account that the pH value is not fixed in the aqueous medium, which can affect the amount of sorption [1], we studied the adsorption of gelatin from water and a phosphate buffer solution, the pH of which is 6.86 and the presence of salts in dissociated form (0.05 M) bring the experiment closer to physiological conditions (model of the upper part of the small intestine). The buffer solution was prepared from a standard mixture of sodium hydrogen phosphate and potassium phosphate according to GOST 8.135:2009.
From fig. 3, it follows that the Langmuir-type adsorption isotherms obtained [7]. For highly dispersed silicon dioxide, the maximum adsorption (Amax) of gelatin from the buffer solution reaches 480 mg/g, and from water only 300 mg/g. The efficiency of adsorption of SAC by highly dispersed silicon dioxide is characterized by the value Amax = 100 mg/g. The difference in gelatin adsorption values when the pH changes is due to the fact that the adsorption of protein molecules is controlled by electrostatic interactions [1,8] and, in addition to pH, also depends on the ionic strength of the solution.
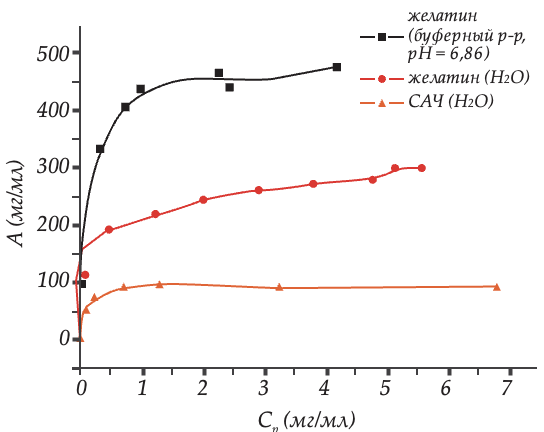
Rice. 3. Adsorption isotherms of gelatin from a buffer solution and water and SACH from water on the surface of highly dispersed silicon dioxide (Atoxyl).
Adsorption of protein molecules on the surface of hydrophilic silica (Atoxyl) is carried out quite quickly. In fig. 4 presents the kinetic curve of gelatin adsorption from water by highly dispersed silicon dioxide. It can be seen that 70 % of the protein present in the solution is absorbed by the silica surface during the first 10 min (200 mg/g) of interaction. After this, the rate of adsorption slows down, and a bend is observed on the kinetic curve, which corresponds to an adsorption time of 15 min (250 mg/g). According to the kinetic curve, it can be concluded that within 30 minutes (280 mg/g) highly dispersed silicon dioxide practically fully realizes its adsorption potential.
Among other investigated sorbents, activated carbon does not adsorb SAC at all, which completely coincides with the results of previous studies [9]. The coal pores are too narrow for protein substances and other high-molecular compounds to penetrate into them. Adsorption of SAC by dioctahedral smectite is insignificant, the adsorption indicators obtained in different experiments are concentrated in the range of 40–60 mg/g. The value of the maximum adsorption of SAC from water for the methylsilicic acid hydrogel is 330 mg/g.
Treatment of aqueous dispersion of highly dispersed silicon dioxide with ultrasound has a significant effect on the amount of gelatin adsorption. After ultrasonic treatment of hydrophilic silica for 5 minutes, the maximum adsorption value increases to 580 mg/g. This is due to the fact that the maximum amount of active silanol groups is released on the surface of silica, which makes them more accessible for the adsorption of protein molecules.
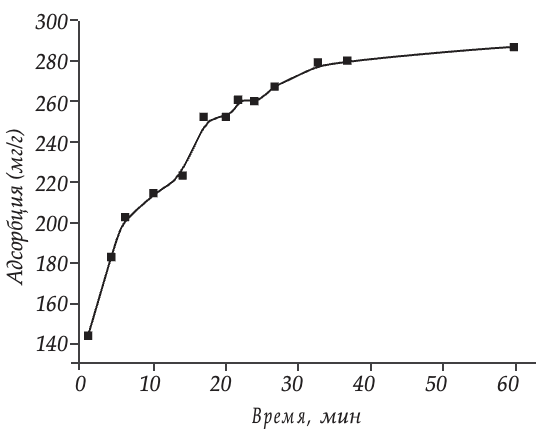
Rice. 4. Kinetics of gelatin adsorption on the surface of highly dispersed silicon dioxide (Atoxyl) from an aqueous solution.
Maximum sorption (Amax, mg/g) of SAC and gelatin by the studied preparations
Table 4.

Table 5.
Maximum absorption (Amax, mg/g) of the studied drugs in terms of a single therapeutic dose.

The adsorption characteristics of highly dispersed silicon dioxide in comparison with the maximum adsorption values obtained for other adsorbents are shown in table. 4.
To compare the therapeutic effect of the studied enterosorbents, we recalculated the adsorption value for a single dose specified in the instructions for the use of the drugs (http://mozdocs.kiev.ua) [10]. The results are shown in the table. 5. As can be seen, the efficiency of sorption with respect to SAC from an aqueous medium increases in the order of dioctahedral smectite < hydrogel of methylsilicic acid < highly dispersed silicon dioxide, and with respect to gelatin both from water and from a buffer solution, it increases in the order of hydrogel of methylsilicic acid < dioctahedral smectite < highly dispersed silicon dioxide.
Thus, with enteral use in the form of ready-made dosage forms presented on the pharmaceutical market of Ukraine, highly dispersed silicon dioxide can be considered the most effective in relation to the sorption of protein compounds.
Conclusions:
- The maximum value of sorption in relation to protein molecules is shown by highly dispersed silicon dioxide at a pH corresponding to the medium of the upper part of the small intestine.
- The rate of adsorption of proteins on the surface of highly dispersed silicon dioxide is quite high. Within 30 minutes, Atoxyl almost fully realizes its adsorption potential.
- Activated carbon practically does not absorb proteins, since its structure is represented by micropores that are inaccessible for the penetration of high-molecular compounds.
- In calculating the amount of protein sorption per therapeutic dose, highly dispersed silicon dioxide (Atoxyl) is significantly superior to methylsilicic acid hydrogel and dioctahedral smectite, which is especially evident in the case of gelatin adsorption.
Literature:
- Lutsyuk M. B., Kulyk Ya. M. Highly dispersed pyrogenic silica as a means of efferent and enterosorption therapy // Saponite and aerosil in livestock and medicine / M. F. Kulyk [et al.]; eds. M. F. Kulyk, T. V. Zasukha, M. B. Lutsyuk. – Vinnytsia: Rogalska I. O., 2012. – 362 p.
- Medicinal chemistry and clinical application of silicon dioxide // Ed. A. A. Chuyko – Kyiv: Naukova dumka, 2003.
- Gerashchenko I. I. Enterosorbents: medicines and dietary supplements, Kyiv: NASU, O. O. Chuyko Institute of Surface Chemistry, 2014. – 248 p.
- Bergna HE (Ed.) Colloidal Silica: Fundamentals and Applications, Taylor & Francis LLC, Salisbury, 2005.
- Yu.V. Ezepchuk Pathogenicity as a function of biomolecules. - M., 1985.
- Preclinical study of enterosorbents. Method. recommendations /V.G. Nikolaev, N.T. Kartel, E.A. Posohova et al. - Kyiv: State Expert Center of the Ministry of Health of Ukraine, 2010. - 56 p.
- Ayler R. Chemistry of silica: trans. with English, Part 1, 2. - M.: Mir, 1982. - 1127 p.
- Gunko V. M., Turov V. V., Horbyk P. P. Water at the interphase boundary. - Kyiv: Nauk. dumka, 2009. -694 p.
- Khubutia M. Sh., Tsivadze A. Yu., Garaeva G. R., Andreev V. N., Goldyn M. M. Adsorption of free hemoglobin by electrochemically modified activated carbons. Message 2. Blood plasma. // Macroheterocycles – 2012. – 5 (4–5). - pp. 327–332.
- Official instructions for the use of drugs (http://mozdocs.kiev.ua).
