Study of the efficacy and safety of the enterosorbent Atoxil for acute intestinal infections in children
S.O. Kramarev, O.A. Dmitrieva
Bogomolets National Medical University, Kyiv
Resume. 60 children aged 1 to 13 years, patients with acute intestinal infections (AII), were examined, 30 of whom received the drug Atoxil during complex therapy. The criteria for assessing the effectiveness of treatment were: the dynamics of reducing symptoms of intoxication, the disappearance of pain in the abdomen, normalization of bowel movements. It was proven that Atoxil is an effective remedy in the complex therapy of acute intestinal infections in children, the drug is well tolerated, does not cause serious adverse reactions. Atoxil can be recommended for the treatment of intestinal infections of various etiologies.
Keywords: children, therapy, acute intestinal infections, Atoxil.
Introduction
In recent years, approaches to etiotropic therapy of acute intestinal infections (AII) in children have been revised. The number of indications for the prescription of antibiotics and other antibacterial drugs has significantly decreased. The reason for this is that among the pathogens of AII, a large number of strains have appeared that are multi-resistant to antibiotics. Some pathogens have acquired the phenomenon of antibiotic dependence and include them in their metabolic processes [2].
Antibiotics suppress the growth of normal intestinal microflora. Against the background of this process, there is an increased growth of antibiotic-resistant opportunistic bacteria. An example of this negative effect of antibiotic therapy for GKI in children is the development of pseudomembranous colitis caused by Cl. difficileIncreased proliferation of opportunistic flora in the body of a patient with GKI leads to its increased excretion into the environment, which makes such patients a source of infection for surrounding children, especially young children.
Antibiotics cause in some cases the destruction of the intestinal mucosa, suppress immunogenesis, and contribute to the formation of lactase deficiency [6, 9].
In addition to the above, the significance of antibacterial therapy for GKI in children is reduced due to the fact that in 50-80% of cases, infectious diarrhea in children is caused either by viruses or anaerobic bacteria that are genetically resistant to antibiotics.
Today, enterosorbents can be an alternative or addition to antibacterial and pathogenetic therapy for acute intestinal infections in children. A large number of enterosorbents are known, differing in particle size, surface features, type of skeleton structure, presence and size of pores, and chemical structure. By chemical structure, enterosorbents are divided into zeolites, inorganic oxides, silica gels, aluminosilicates, organominerals, organic polymers, and activated carbon.
The basis for the use of enterosorbents in the treatment of GI in children is that they are able to fix hundreds of millions of GI causative cells on their surface [4]. It has been established that many sorbents inhibit the adhesion of microorganisms on the surface of the intestinal mucosa, reduce the translocation of microflora from the intestine into the internal environment of the body and, thus, prevent the generalization of the infectious process. Microbes adhered to the surface of enterosorbents are destroyed and excreted from the patient's body [1, 3, 4]. Together with bacterial pathogens of GI, enterosorbents fix rotaviruses located in the intestinal cavity on their surface [8]. In addition to pathogens of the disease, enterosorbents remove microbial toxins and products of their metabolism from the body. They act as activators of catalytic processes in the body and transform toxic substances into low-toxic ones [5].
The most promising in the treatment of GKI in children are silicon enterosorbents, which in terms of their activity surpass many other enterosorbents and are safe for children. Unlike coal sorbents, they do not require the administration of large volumes of the drug to achieve a therapeutic effect, and are significantly better in terms of organoleptic properties. The presence of micropores in coal enterosorbents prevents the sorption of high-molecular protein toxins that accumulate in the body during infectious toxicosis [5]. Also, coal sorbents penetrate the submucosal layer of the intestine and can damage it [7].
The pharmaceutical market of Ukraine presents the enterosorbent Atoxil, which is silicon dioxide and has the largest active sorption area (over 400 m² per 1 g of substance). It has pronounced sorption properties, exhibits detoxification, antimicrobial effects, adsorbs from the digestive tract and removes endogenous and exogenous toxins of various origins from the patient's body, including food and bacterial allergens, microbial endotoxins and other toxic substances, toxic products formed during the breakdown of proteins in the intestine.
Atoxil promotes the transport of various toxic products from the internal environment of the body (blood, lymph, interstitium) into the digestive tract due to concentration and osmotic gradients, including medium molecules, oligopeptides, amines and other substances with their subsequent removal from the body. The drug is practically not absorbed in the digestive tract, has pleasant organoleptic properties, which allow its wide use in pediatric practice.
Research material and methods
The study involved 60 children with acute intestinal infections, aged 1 to 13 years, including 30 children (main group) who received Atoxil during complex therapy, and 30 patients (control group) who were treated with generally accepted methods for this disease. All children had a moderately severe form of the disease.
Basic GCI therapy in both groups was carried out according to the “Protocols for the diagnosis and treatment of infectious diseases in children” (Order of the Ministry of Health of Ukraine No. 354 dated 09.07.04).
The exclusion criteria from the study were: a history of hypersensitivity to the components of the drug and the presence of concomitant serious diseases.
Table 1
Therapeutic doses of Atoxil based on the child's weight
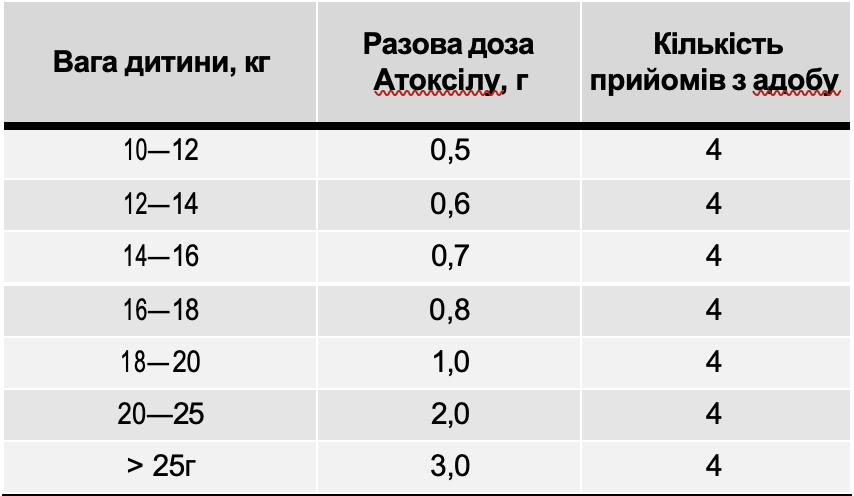
The daily dose of Atoxil for children aged 7 years and older was 12 g (if necessary, it can be increased to 24 g); for children aged 1 to 7 years — 150–200 mg per 1 kg of body weight. The daily dose of Atoxil was divided into 3–4 doses. The maximum single dose of the drug should not exceed half the daily dose. The course of treatment was 5 days. The children took the aqueous suspension of the drug 1 hour before taking food or medications (Table 1).
Table 2 shows data on the distribution of children taking into account the etiology of intestinal infections (based on the results of bacteriological examination of feces).
Tables 3 and 4 present data on the distribution of children by gender and age.
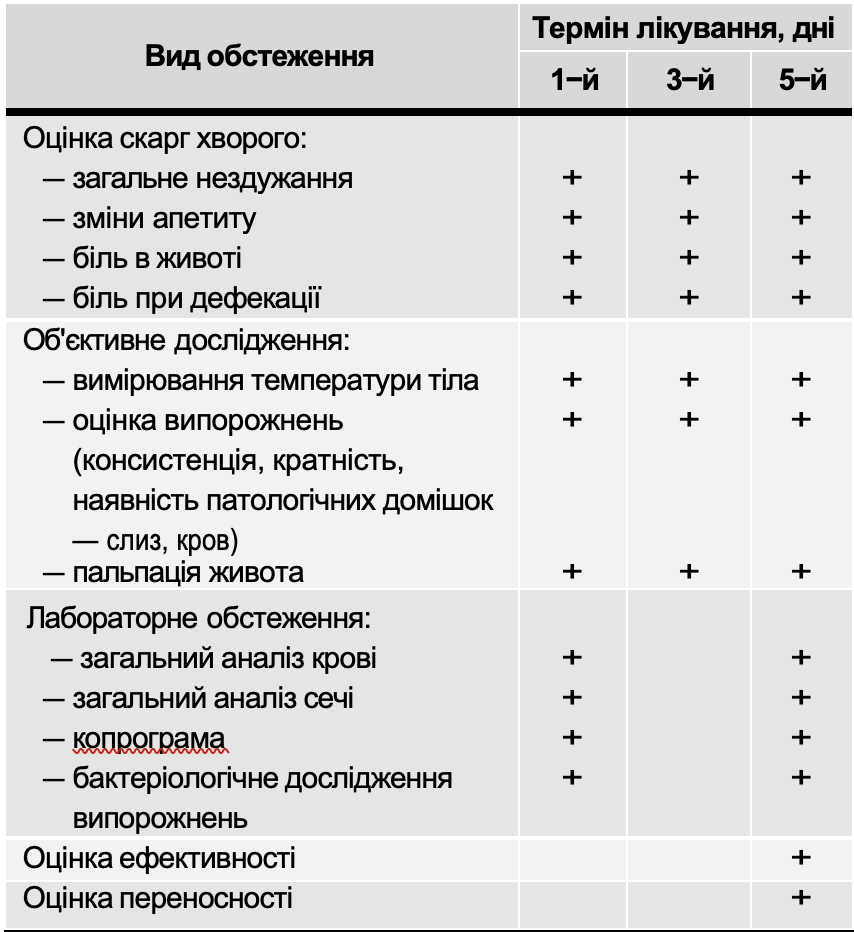
All patients included in the study were examined using the following clinical and laboratory methods (Table 5).
Table 2
Distribution sick children by etiology of intestinal infections
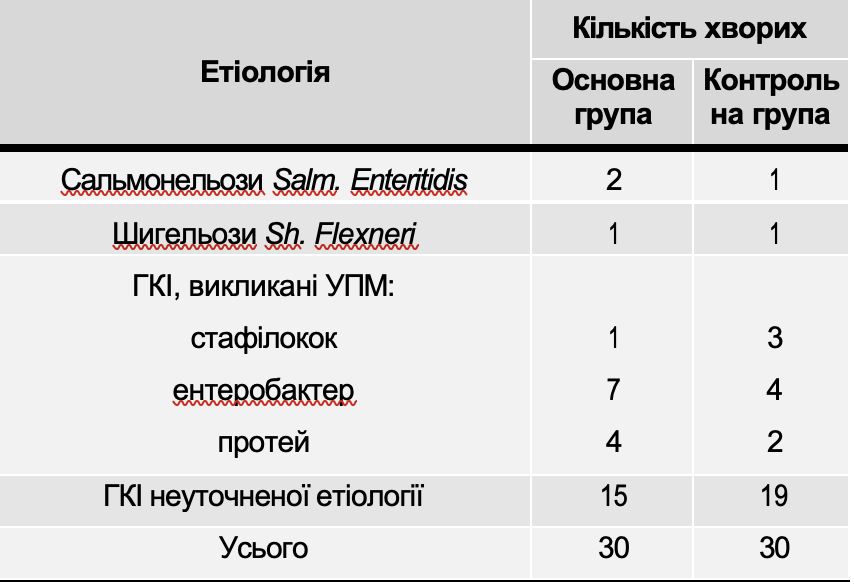
Table 3
Distribution children from GKI by article

The criteria for evaluating the effectiveness of treatment were:
- dynamics of reducing symptoms of intoxication;
- dynamics of the disappearance of abdominal pain;
- dynamics of normalization of stools (consistency, frequency, presence of pathological impurities). The data used as criteria for the effectiveness of therapy were evaluated according to a verbal scale and expressed in points: 0 points - the symptom is not pronounced, 1 point - the severity of the symptom is moderate, 2 points - the symptom is pronounced. Based on the results obtained, a conclusion was made about the effectiveness of the use of the drug Atoxil.
Examinations performed by children with GKI
Table 5
The degree of treatment effectiveness was assessed using criteria that monitored the level of intoxication of the body.
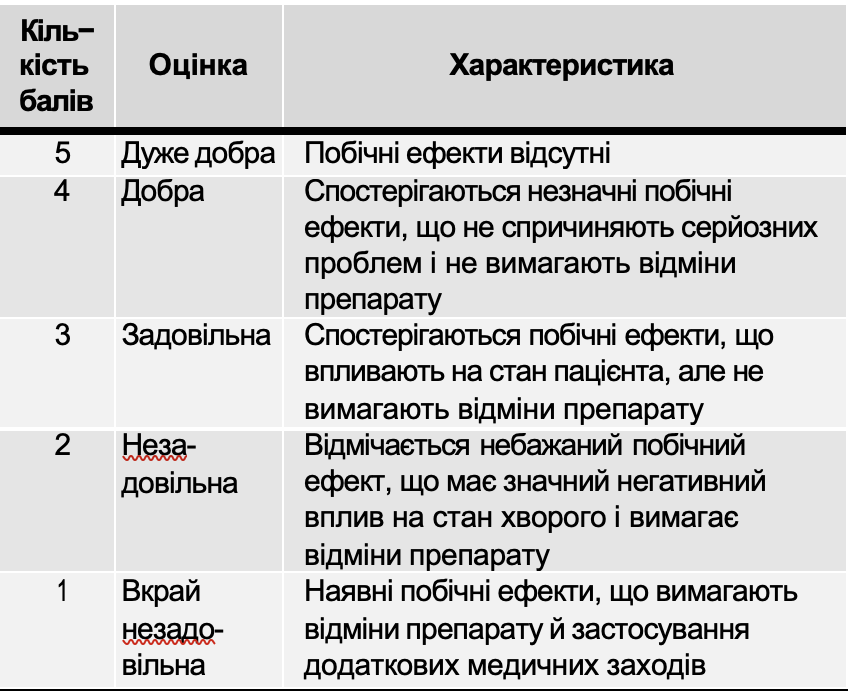
The tolerability of the drug was assessed based on subjective symptoms and objective data obtained by the investigator during treatment. The dynamics of laboratory parameters, as well as the frequency and nature of side effects, were taken into account. The tolerability of the drug was assessed in points (Table 7).
When using Atoxil as part of complex therapy, a causal relationship between side effects and the study drug was established.
Table 6
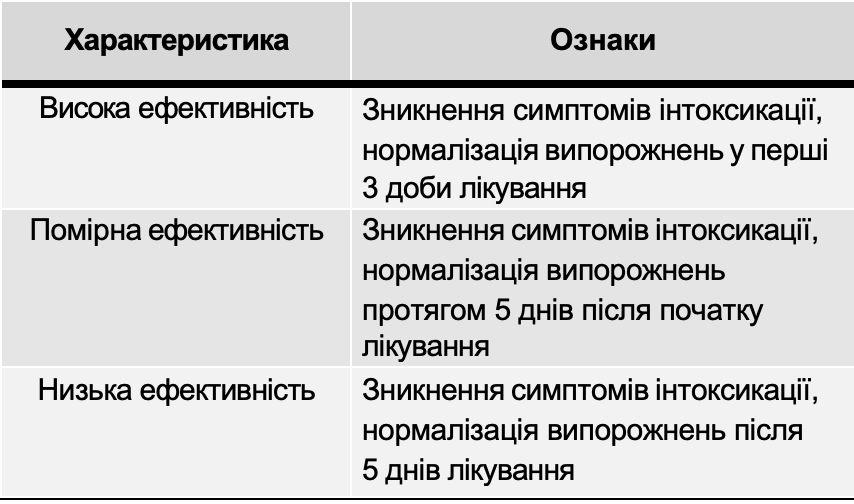
Table 7
Assessment of side effects on the body of the drug Atoxil
Research results and their discussion
Table 8 presents an assessment of the dynamics of clinical symptoms of GKI in patients of the main and control groups.
As can be seen from Table 8, on the first day of treatment, symptoms of general malaise, decreased appetite, headache, and abdominal pain were observed in all patients of both the main and control groups. In 34 children of both groups, symptoms of intoxication were moderately expressed, in 26 - their significant severity was noted. On the third day of treatment, these symptoms were not observed in 19 patients of the main and 17 of the control groups; in 24 sick children of both groups, their moderate severity was noted. On the fifth day of treatment, almost all patients of both groups felt satisfactorily.
An increase in body temperature on the first day was noted in 57 studied patients (in most cases it was within 37-38°, only in 17 patients of both groups it was higher than 38°). On the third day of observation, body temperature was normal in 36 children (17 - main group, 19 - control group). Five days after hospitalization, almost all patients had a temperature within 36-37°.
During the clinical examination of patients, an indicator such as abdominal pain was taken into account. This symptom on the first day of observation was moderately expressed in 53 patients. On the third day from the start of treatment in the main group of children, abdominal pain was no longer observed in 14 patients, and was moderately expressed in 16 examined patients. In the control group of children, this symptom was absent only in 11 patients, and in 19 it was moderately expressed. On the fifth day of treatment, abdominal pain was not observed in any patient.
In addition to symptoms of general intoxication, all patients were assessed for local symptoms of gastrointestinal tract damage. Stools were assessed (their frequency, consistency, and the presence of pathological impurities in the form of mucus and blood).
The frequency of bowel movements in the acute period of the disease in all children was increased. In 30 patients of both groups it was up to 5 times a day, in 30 patients - more than 5 times. On the third day of treatment in the main group in 15 patients the frequency of bowel movements was 1-2 times a day, in 13 - up to 5 times. In the control group during this period this indicator was within the physiological norm in only 10 children. On the fifth day from the start of treatment in 28 children who received the drug Atoxil, the frequency of bowel movements was 1-2 times a day, while in the control group only in 20 children it was normal (p < 0.05).
All patients under observation had a violation of stool consistency on the first day of treatment. In the vast majority of children (56 patients in both groups), they were loose. In the dynamics of the disease, stool gradually normalized, but in the group of children receiving the drug Atoxil, this process occurred significantly faster (p < 0.05). Three days after the start of treatment, only 9 children in the main group had loose stools, in 7 children they had a formed appearance, while in the control group, only one patient had normal stool consistency. On the fifth day of treatment in the main group, stool normalized in almost all patients.
Of the pathological impurities, the presence of mucus in the feces was noted in most patients. Blood impurities in the form of streaks were observed only in 5 sick children of both groups. In the dynamics of the disease, pathological impurities in the feces gradually disappeared. At the end of treatment, blood impurities were not noted in any patient, mucus in a small amount was only in 3 children (in 1 patient of the main group and 2 - control).
Fig. 1 shows the frequency of bowel movements in children of the main and control groups on the 1st, 3rd and 5th days of treatment.
Fig. 2 shows the dynamics of normalization of stool consistency in sick children of the main and control groups.
All patients underwent laboratory examinations: complete blood count, complete urine analysis, coprological and bacteriological examination of feces. Laboratory examinations were performed twice: at the beginning and at the end of treatment.
When analyzing blood parameters, abnormalities were noted that are characteristic of the acute period of intestinal infections: the number of erythrocytes and the level of hemoglobin were almost the same as the physiological norm. The total number of leukocytes in the majority of patients was increased, an increase in neutrophils was noted in the leukocyte formula of the blood, the ESR was slightly increased. During the examination, the blood parameters normalized in all examined patients.
Urinalysis results were normal in all children. Stool analysis revealed signs of small or large intestinal involvement.
Table 8
Dynamics of main clinical symptoms during treatment
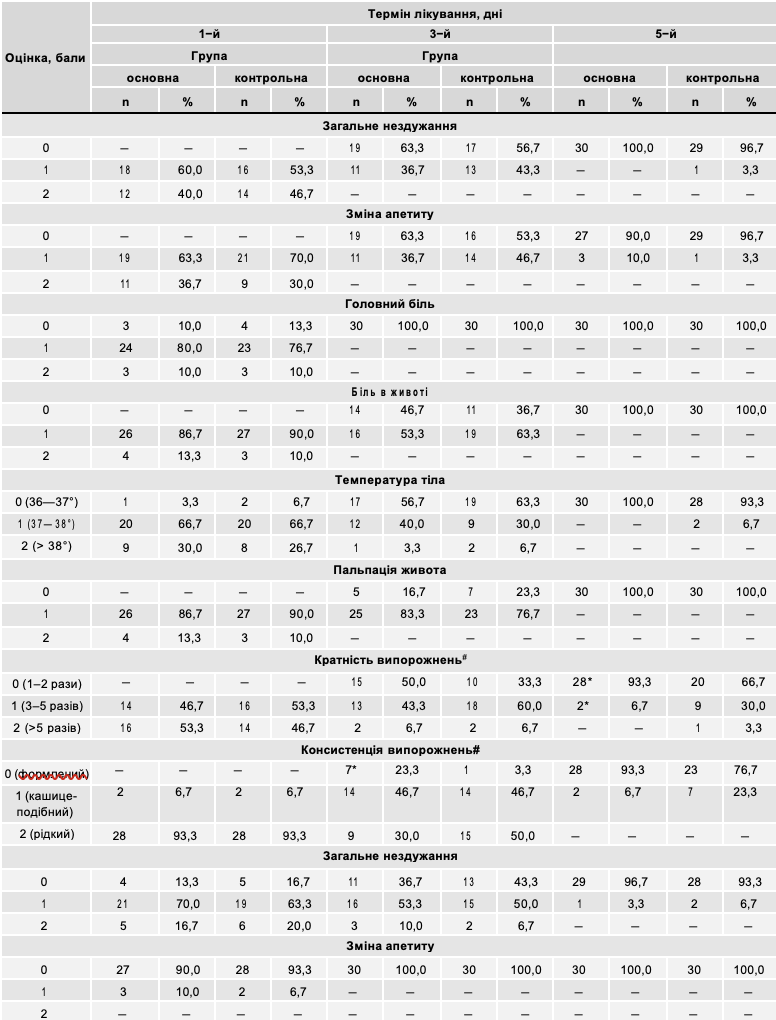
Note. * — significant difference compared to the control group indicator using the method of assessing the difference between the frequencies of the appearance of a symptom in individual observation series (p<0.05);
# — significant effect of treatment with Atoxil on the dynamics of the symptom according to two-factor analysis of variance (p<0.05).
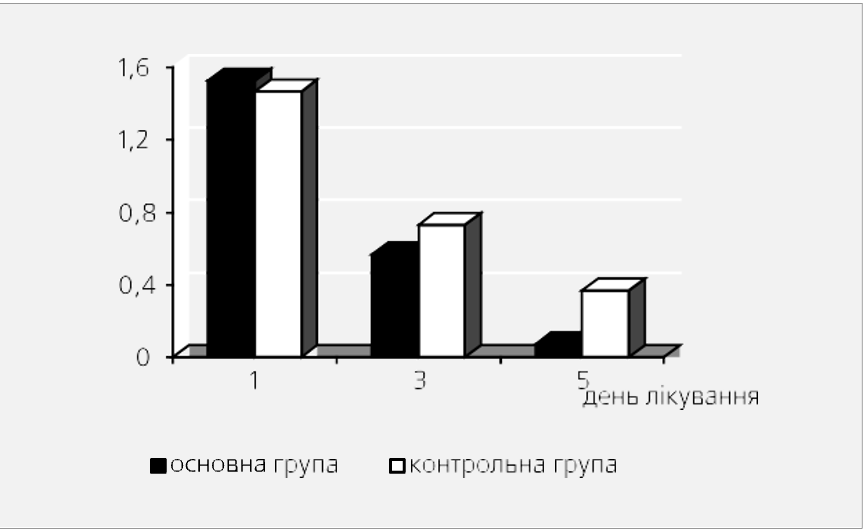
Fig. 1. Dynamics multiplicity bowel movements at different methods therapy (average score).
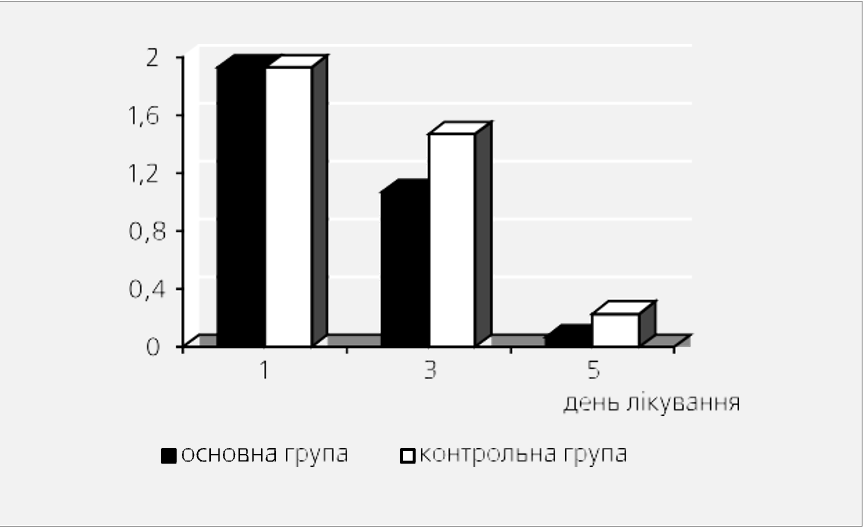
Fig. 2. Dynamics of stool consistency with different therapy methods (average score).
Table 9
Evaluation of the effectiveness of GCS therapy using Atoxil
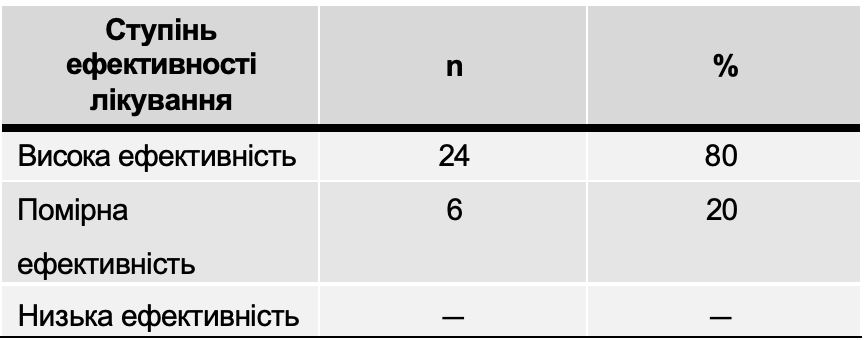
Table 10
Registration side effects effects drug Atoxil
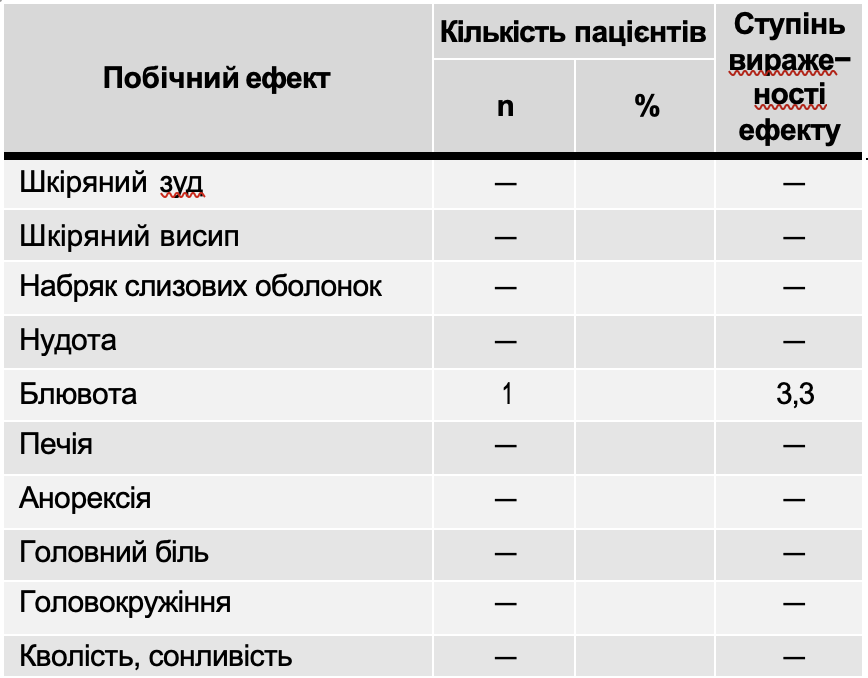
Note. + – mild side effect; ++ – moderate side effect; +++ – severe side effect.
During the control examination after treatment of children in whom pathogenic microorganisms were isolated during bacteriological examination of feces, complete sanitation was noted (in 100% patients in both groups).
The results obtained indicate the high therapeutic effectiveness of the use of the drug Atoxil in the complex therapy of acute intestinal infections in children.
Table 9 presents the results of assessing the degree of effectiveness of treatment with the drug Atoxil. In all children, if side effects occurred due to the use of Atoxil, they were registered and their severity was determined (Table 10).
As can be seen from Table 10, side effects of the drug Atoxil in the form of vomiting were noted in 1 (3.3%) patient. In this case, a break in taking the drug was made for 10-12 hours (1-2 doses), after which the use of Atoxil was resumed in the same doses; these side effects did not require discontinuation of the drug.
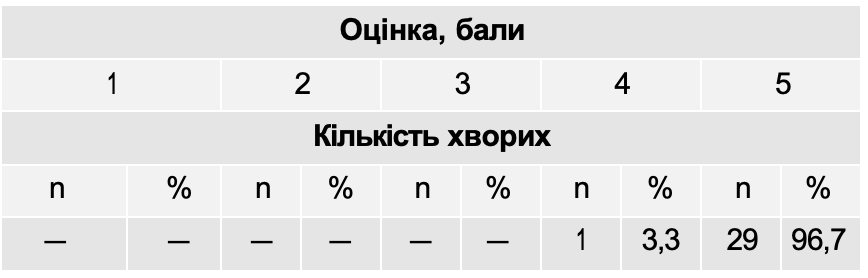
The results of the assessment of the tolerability of the drug Atoxil are presented in Table 11.
Table 11
Assessment of the tolerability of the drug Atoxil
Conclusions
- Atoxil is an effective treatment in the complex therapy of acute intestinal infections in children.
- The drug Atoxil is well tolerated and does not cause serious side effects.
- The drug Atoxil can be recommended for inclusion in the complex therapy of acute intestinal infections in children of various etiologies.
LITERATURE
- Almagobetov K.H., Gorskaya E.M. Translocation of intestinal flora and its mechanisms // // Microbiology, epidemiology and immunology. — — No. 1. — P. 74-79.
- Vasiliev V.S., Komar V.I., Tsyrkunov V.M. Practice of an infectious disease specialist. — Minsk: Higher School, 1993.
- Vorotyntseva N.V., Gorelova A.V., Tarkovskaya V.A. Clinical and experimental substantiation of enterosorbent "Smekta" in complex therapy of acute intestinal infections in children // Pediatrics. — — No. 7-9. - pp. 46-48.
- Grigoriev A.V., Cartel N.T. Adhesion pathogenic microflora on carbon sorbents// Microbiology, epidemiology and immunology. — 1991. — No. 7. — P. 11-14.
- Eretskaya E.V., Sakhno L.A., Myrutenko N.V. Sorption methods detoxification in medicine. — Kharkov, 1992.
- Ishchuk I.S. Clinic and diagnosis of acute intestinal diseases with disaccharide insufficiency: Author's review. thesis Ph.D. honey. of science — K., 1986.
- Nikolayeva L.G. Experimental study of the effectiveness of enterosorbents in salmonellosis // Microbiology, epidemiology and immunology. — 1994. —No. 2. — pp. 40-44.
- Ray WITH. Viral diarrhea. Experience use smectite // Inter. Review of Pediatrics. — 1989. — No. 196. — P. 1-4.
- Shuvalova E.P., Antonova G.V., Varnovskaya V.V. Polyphepan enterosorbent for salmonellosis and non-specific ulcerative colitis // Terapev. archive — 1991.— No. 11. — WITH. 47-49.
STUDY EFFICIENCY I APPLICATION SAFETYENTEROSORBENT ATOXYL AT ACUTE INTESTINAL INFECTIONS IN CHILDREN
S.O. Kramarev, O.A. Dmitrieva
Resume. 60 children aged from 1 to 13 years with acute intestinal infections were examined, 30 of whom received enterosorbent Atoxyl during complex therapy. The criteria for evaluating the effectiveness of the drug were the dynamics of the reduction of intoxication symptoms, the disappearance of pain in the abdomen, and the normalization of the stool. It has been proven that Atoxyl is an effective remedy for the complex therapy of acute intestinal infections in children, the drug has good tolerance and does not cause serious side effects. Atoxyl can be recommended for the treatment of intestinal infections of various etiologies.
Keywords: children, therapy, acute intestinal infections, Atoxyl.
